
Another exception to general characteristics of metals is alkali metals such as lithium, sodium, potassium which are so soft that they can be cut with a knife. The melting points of metals are usually high but gallium and caesium are those metals having such low melting points that they will melt if you keep them on your palm. At room temperature, most of the metals are solids and have a characteristic silvery shine (except for mercury, which is a liquid). 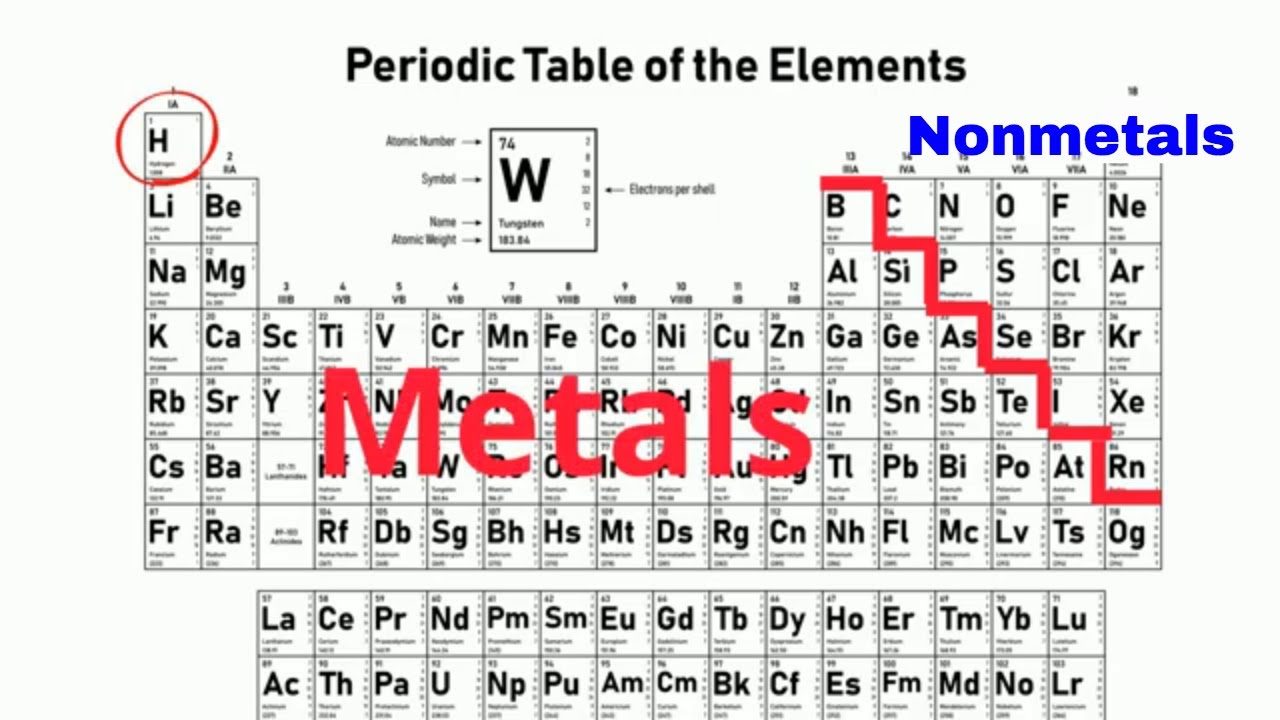
Metals being ductile can be drawn into wire. Metals also possess malleability, that is they can be hammered into sheets. Metals are good conductors of heat and electricity. To the right of the line lie nonmetals, the metals are to the left of the line(except hydrogen, which is a nonmetal), and the elements which lie immediately adjacent to the line are the metalloids.Ī metal is generally a hard solid substance that has a shiny luster and it conducts heat and electricity. This type of classification is based on the shared physical and chemical properties.Ī jagged black line in a periodic table (see figure below) along the right side of the table separates the metals from the nonmetals. Metals and non-metals are generally heard of but here we will also study the intermediate type of elements called metalloids. The elements have one form of classification where they are broadly classified as metals, nonmetals, and metalloids. It is a basic step before we enter the chapter and read about the elements in detail, so the periodic table gives us a brief idea about the elements in the shortest form possible. The arrangement and structure of the periodic table makes it easier and accessible for us to see the link and relationship between the elements at a glance, it is helpful in quickly locating the elements and in turn attach our own heuristics to learn the table as well. Periodic table helps us to identify those elements based on their property which can also indicate to us their weight, whether it is a normal element or comes under noble category etc. The periodic table then places or lists all the known elements together with the similar properties and in a sequential manner. Each element, which is about 100, has its own type of atom because every matter contains atoms and then ultimately everything in the universe contains at least one or more elements. The most important characteristic of an element is that it cannot be broken down into any other substance and that is why it is written that it is extracted in its original form. How do we figure out the position of elements in the periodic table? Elements are natural extracts that we yield naturally and some are made synthetically but the original form has to be extracted no matter what. Mendeleev's version placed and ordered the elements according to increasing atomic weight rather than atomic number. 
The modern table is derived from Mendeleev's form of the table, but has significant differences. The Russian scientist Dmitri Mendeleev is credited with inventing the periodic table (1869). The tabular arrangement of the chemical elements by increasing atomic number to see the trends in their properties is called a periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
